How Many Electrons In Krypton
Crystal Structure of Krypton
The solid country structure of Krypton is Face Centered Cubic.
The Crystal construction can be described in terms of its unit Cell. The unit Cells repeats itself in three dimensional space to form the structure.
Unit Cell Parameters
The unit cell is represented in terms of its lattice parameters, which are the lengths of the cell edges Lattice Constants (a, b and c)
| a | b | c |
|---|---|---|
| 570.6 pm | 570.half-dozen pm | 570.6 pm |
and the angles betwixt them Lattice Angles (alpha, beta and gamma).
| alpha | beta | gamma |
|---|---|---|
| π/ii | π/2 | π/2 |
The positions of the atoms inside the unit jail cell are described by the ready of atomic positions ( xi, yi, zi) measured from a reference lattice point.
The symmetry properties of the crystal are described by the concept of space groups. All possible symmetric arrangements of particles in 3-dimensional space are described past the 230 space groups (219 distinct types, or 230 if chiral copies are considered distinct.
Krypton Atomic and Orbital Properties
Krypton atoms have 36 electrons and the electronic shell structure is [ 2, viii, eighteen, 8 ] with Atomic Term Symbol (Breakthrough Numbers) 1S0.
Bohr Atomic Model of Krypton - Electrons per energy level

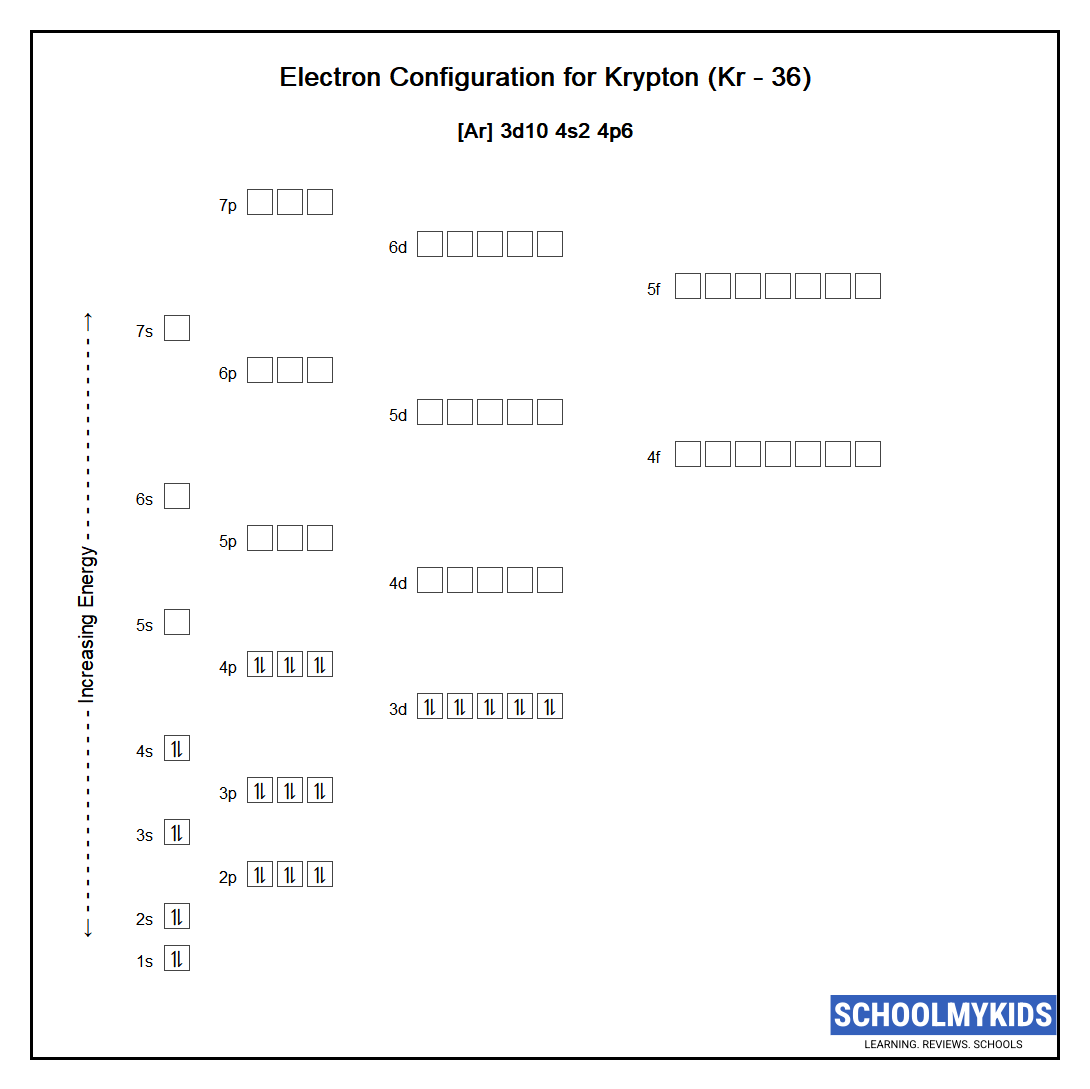
Footing State Electronic Configuration of Krypton - neutral Krypton atom
Abbreviated electronic configuration of Krypton
The footing country abbreviated electronic configuration of Neutral Krypton cantlet is [Ar] 3d10 4s2 4p6 . The portion of Krypton configuration that is equivalent to the noble gas of the preceding flow, is abbreviated equally [Ar] . For atoms with many electrons, this note can become lengthy then an abbreviated notation is used. This is of import as it is the Valence electrons 4s2 4p6 , electrons in the outermost shell that make up one's mind the chemical properties of the element.
Unabbreviated electronic configuration of neutral Krypton
Complete footing country electronic configuration for the Krypton atom, Unabbreviated electronic configuration
1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6
Electrons are filled in atomic orbitals as per the order determined past the Aufbau principle, Pauli Exclusion Principle and Hund'southward Rule.
Atomic Construction of Krypton
Krypton atomic radius is 88 pm , while information technology's covalent radius is 110 pm .
| Atomic Radius Calculated | 88 pm ( 0.88 Å) |
| Atomic Radius Empirical | - ( 0 Å) |
| Diminutive Book | 22.35 cm3/mol |
| Covalent Radius | 110 pm ( i.1 Å) |
| Van der Waals Radius | 202 pm |
| Neutron Cross Section | 25 |
| Neutron Mass Absorption | 0.013 |
Diminutive Spectrum of Krypton
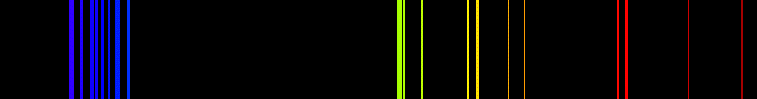
Krypton Chemical Properties: Krypton Ionization Energies and electron analogousness
The electron analogousness of Krypton is 0 kJ/mol .
Ionization Free energy of Krypton
Refer to table below for Ionization energies of Krypton
| Ionization energy number | Enthalpy - kJ/mol |
|---|---|
| 1st | 1350.8 |
| second | 2350.4 |
| 3rd | 3565 |
| 4th | 5070 |
| fifth | 6240 |
| 6th | 7570 |
| seventh | 10710 |
| 8th | 12138 |
| 9th | 22274 |
| 10th | 25880 |
| 11th | 29700 |
| 12th | 33800 |
| 13th | 37700 |
| 14th | 43100 |
| 15th | 47500 |
| 16th | 52200 |
| 17th | 57100 |
| 18th | 61800 |
| 19th | 75800 |
| 20th | 80400 |
| 21st | 85300 |
| 22nd | 90400 |
| 23rd | 96300 |
| 24th | 101400 |
| 25th | 111100 |
| 26th | 116290 |
| 27th | 282500 |
| 28th | 296200 |
| 29th | 311400 |
| 30th | 326200 |
Krypton Physical Properties
Refer to below tabular array for Krypton Concrete Properties
| Density | 0.00375 g/cm3 |
| Tooth Volume | 22.35 cm3/mol |
Elastic Backdrop
Hardness of Krypton - Tests to Measure of Hardness of Element
Krypton Electrical Properties
Krypton is Conductor of electricity. Refer to tabular array beneath for the Electric backdrop of Krypton
Krypton Heat and Conduction Properties
Krypton Magnetic Properties
Optical Properties of Krypton
Acoustic Properties of Krypton
Krypton Thermal Properties - Enthalpies and thermodynamics
Refer to table below for Thermal properties of Krypton
Enthalpies of Krypton
Krypton Isotopes - Nuclear Properties of Krypton
Krypton has 32 isotopes, with betwixt 69 and 100 nucleons. Krypton has 6 stable naturally occuring isotopes.
Isotopes of Krypton - Naturally occurring stable Isotopes: 78Kr, 80Kr, 82Kr, 83Kr, 84Kr, 86Kr .
| Isotope | Z | N | Isotope Mass | % Abundance | T half | Decay Manner |
|---|---|---|---|---|---|---|
| 69Kr | 36 | 33 | 69 | Synthetic | ||
| 70Kr | 36 | 34 | seventy | Synthetic | ||
| 71Kr | 36 | 35 | 71 | Synthetic | ||
| 72Kr | 36 | 36 | 72 | Synthetic | ||
| 73Kr | 36 | 37 | 73 | Synthetic | ||
| 74Kr | 36 | 38 | 74 | Constructed | ||
| 75Kr | 36 | 39 | 75 | Constructed | ||
| 76Kr | 36 | twoscore | 76 | Synthetic | ||
| 77Kr | 36 | 41 | 77 | Synthetic | ||
| 78Kr | 36 | 42 | 78 | 0.35% | Stable | Northward/A |
| 79Kr | 36 | 43 | 79 | Synthetic | ||
| 80Kr | 36 | 44 | fourscore | ii.28% | Stable | North/A |
| 81Kr | 36 | 45 | 81 | Synthetic | ||
| 82Kr | 36 | 46 | 82 | 11.58% | Stable | North/A |
| 83Kr | 36 | 47 | 83 | xi.49% | Stable | Northward/A |
| 84Kr | 36 | 48 | 84 | 57% | Stable | |
| 85Kr | 36 | 49 | 85 | Synthetic | ||
| 86Kr | 36 | fifty | 86 | 17.3% | Stable | N/A |
| 87Kr | 36 | 51 | 87 | Synthetic | ||
| 88Kr | 36 | 52 | 88 | Synthetic | ||
| 89Kr | 36 | 53 | 89 | Synthetic | ||
| 90Kr | 36 | 54 | 90 | Synthetic | ||
| 91Kr | 36 | 55 | 91 | Constructed | ||
| 92Kr | 36 | 56 | 92 | Synthetic | ||
| 93Kr | 36 | 57 | 93 | Constructed | ||
| 94Kr | 36 | 58 | 94 | Synthetic | ||
| 95Kr | 36 | 59 | 95 | Constructed | ||
| 96Kr | 36 | 60 | 96 | Synthetic | ||
| 97Kr | 36 | 61 | 97 | Synthetic | ||
| 98Kr | 36 | 62 | 98 | Synthetic | ||
| 99Kr | 36 | 63 | 99 | Constructed | ||
| 100Kr | 36 | 64 | 100 | Synthetic |
How Many Electrons In Krypton,
Source: https://www.schoolmykids.com/learn/periodic-table/kr-krypton
Posted by: potterlond1937.blogspot.com


0 Response to "How Many Electrons In Krypton"
Post a Comment